There are four laws of thermodynamics – zeroth law, first law, second law and third law. First law of thermodynamics has been explained in another article. Second law of thermodynamics has three different statements – Kelvin-Planck’s statement, Clausius statement and the statement in terms of entropy of universe. In this article, we are going to discuss all these statements along with their explanations.
Kelvin-Planck’s statement for second law of thermodynamics
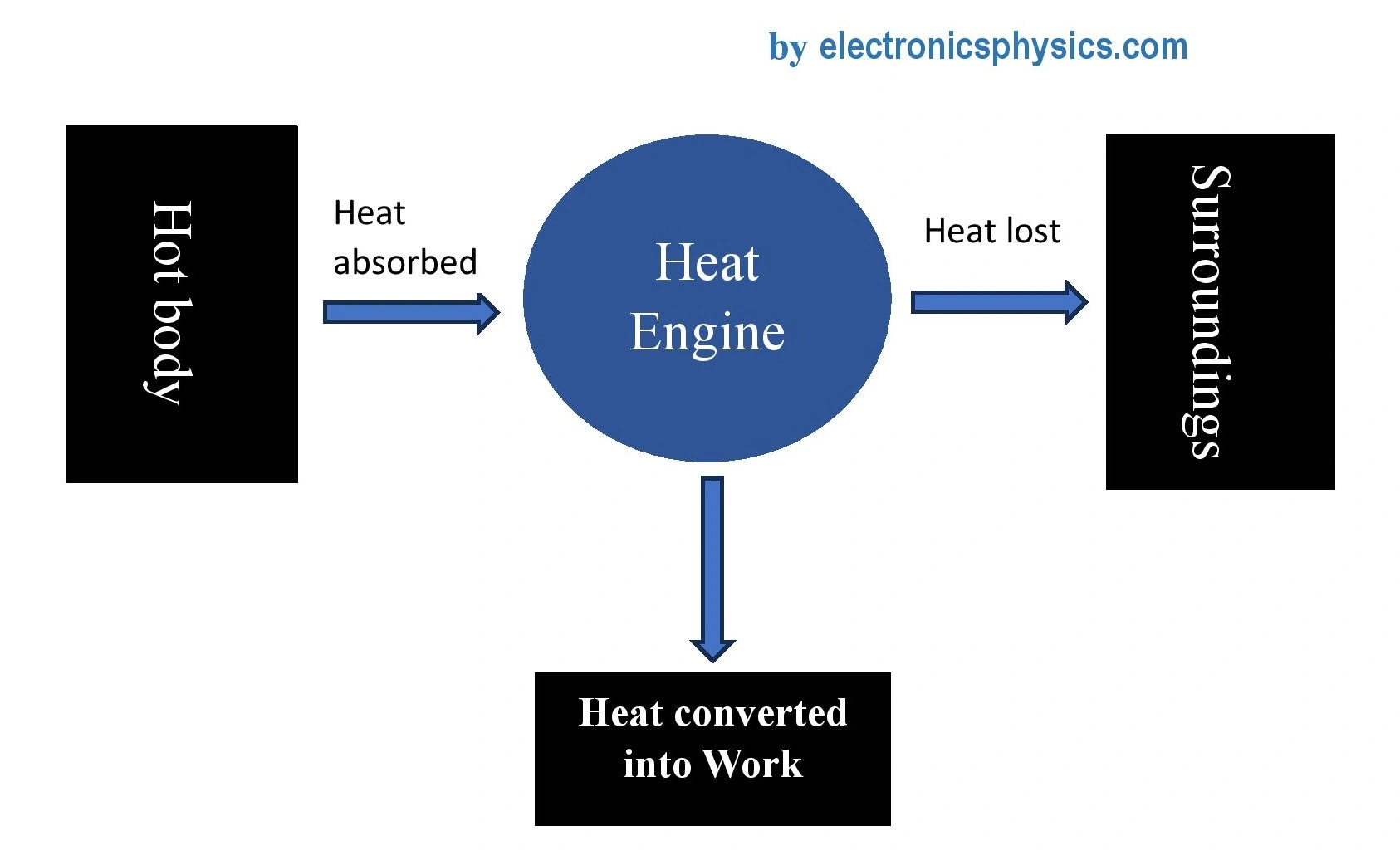
According to Kelvin-Planck’s statement, “no thermodynamic cyclic process is possible which can convert the absorbed heat entirely into work”. This explains that the system losses some of the absorbed heat to its surroudings. For example, if a system absorbs 100 Joules of heat, it cannot do work by 100 Joules, it will do work by less than 100 Joules. This makes the efficiency of the thermodynamic system less than 100%. This concept is useful in heat engines.
Clausius statement for second law of thermodynamics
According to Clausius statement, “no thermodynamic cyclic process is possible which can transfer heat from cold reservior to hot reservior without any external work done”. We know that heat generally flows from hot body to cold body. Now if we want to transfer heat from cold body to hot body, it will not happed sponteneously, rather external work done on the system is required. This concept is useful in refrigerators where we use to cool the cold body further.
Second law of thermodynamics in terms of entropy
The importance of 2nd law of thermodynamics is that it gives the concept of entropy. In fact, this law can be stated in terms of entropy. All natural phenomena are irreversible in nature and entropy is the measure of disorderness or irreversibility of a process executed by a system. Second law of thermodynamics in terms of entropy states that entropy of universe increases in all natural phenomena and reaching towards the maximum entropy value.
Important questions from 2nd law of Thermodynamics
- State 2nd law of Thermodynamics.
- What is the physical significance of 2nd law of thermodynamics?
- State second law of thermodynamics in terms of entropy.
This is all from this article on all three statements for 2nd law of thermodynamics. Hope you understand all three statements. If you have any doubt ask me in the comments. Thank you.
Related article:
1 thought on “Second law of Thermodynamics in terms of entropy”